Longer term effects of very low energy diet on obstructive sleep apnoea in cohort derived from randomised controlled trial: prospective observational follow-up study
BMJ 2011; 342 doi: https://doi.org/10.1136/bmj.d3017 (Published 01 June 2011) Cite this as: BMJ 2011;342:d3017- Kari Johansson, PhD student1,
- Erik Hemmingsson, postdoctoral research fellow1,
- Richard Harlid, physician2,
- Ylva Trolle Lagerros, physician13,
- Fredrik Granath, statistican3,
- Stephan Rössner, professor1,
- Martin Neovius, associate professor3
- 1Obesity Unit, Department of Medicine (Huddinge), Karolinska Institute, SE-141 86 Stockholm, Sweden
- 2Aleris Fysiologlab, Stockholm
- 3Clinical Epidemiology Unit, Department of Medicine (Solna), Karolinska Institute, Stockholm
- Correspondence to: K Johansson kari.johansson{at}ki.se
- Accepted 7 April 2011
Abstract
Objective To determine whether initial improvements in obstructive sleep apnoea after a very low energy diet were maintained after one year in patients with moderate to severe obstructive sleep apnoea.
Design Single centre, prospective observational follow-up study.
Setting Outpatient obesity clinic in a university hospital in Stockholm, Sweden.
Participants 63 men aged 30-65 with body mass index (BMI) 30-40 and moderate to severe obstructive sleep apnoea defined as an apnoea-hypopnoea index ≥15 (events/hour), all treated with continuous positive airway pressure.
Intervention A one year weight loss programme, consisting of an initial very low energy diet for nine weeks (seven weeks of 2.3 MJ/day and two weeks of gradual introduction of normal food) followed by a weight loss maintenance programme.
Main outcome measure Apnoea-hypopnoea index, the main index for severity of obstructive sleep apnoea. Data from all patients were analysed (baseline carried forward for missing data).
Results Of 63 eligible patients, 58 completed the very low energy diet period and started the weight maintenance programme and 44 completed the full programme; 49 had complete measurements at one year. At baseline the mean apnoea-hypopnoea index was 36 events/hour. After the very low energy diet period, apnoea-hypopnoea index was improved by −21 events/hour (95% confidence interval −17 to −25) and weight by −18 kg (−16 to −19; both P<0.001). After one year the apnoea-hypopnoea index had improved by −17 events/hour (−13 to −21) and body weight by −12 kg (−10 to −14) compared with baseline (both P<0.001). Patients with severe obstructive sleep apnoea at baseline had greater improvements in apnoea-hypopnoea index (−25 events/hour) compared with patients with moderate disease (−7 events/hour, P<0.001). At one year, 30/63 (48%, 95% confidence interval 35% to 60%) no longer required continuous positive airway pressure and 6/63 (10%, 2% to 17%) had total remission of obstructive sleep apnoea (apnoea-hypopnoea index <5 events/hour). There was a dose-response association between weight loss and apnoea-hypopnoea index at follow-up (β=0.50 events/kg, 0.11 to 0.88; P=0.013).
Conclusion Initial improvements in obstructive sleep apnoea after treatment with a very low energy diet can be maintained after one year in obese men with moderate to severe disease. Those who lose the most weight or have severe sleep apnoea at baseline benefit most.
Trial registration Current Controlled Trials 70090382.
Introduction
Obstructive sleep apnoea is common, underdiagnosed, and associated with negative health effects, including impaired cognitive function, increased risk for vehicle crashes and occupational injuries, decreased quality of life, and increased mortality.1 2 3 4 5
Of all people with obstructive sleep apnoea, an estimated 60-70% are either overweight or obese.6 7 Given the close association between obstructive sleep apnoea and obesity, weight loss has been advocated as a primary treatment option in obese patients with sleep apnoea. Data from randomised controlled trials supporting this concept were lacking until 2009, when three trials on the effects of weight loss in obstructive sleep apnoea were published.8 9 10 Tuomilehto et al investigated the effect of a very low energy diet followed by supervised lifestyle counselling for one year in overweight or obese patients with mild obstructive sleep apnoea.8 Foster et al investigated the effects of intensive lifestyle changes in overweight or obese patients with type 2 diabetes and mild to severe obstructive sleep apnoea for one year.9 Finally, we investigated the effects of very low energy diet for nine weeks in obese men with moderate to severe obstructive sleep apnoea,10 the severity of disease associated with an increased risk of mortality.3 4 11 All three trials found that weight loss resulted in clinically relevant improvements in the apnoea-hypopnoea index, the main index for severity of obstructive sleep apnoea.
Despite an improving case for the robust treatment effect of weight loss in obstructive sleep apnoea, concerns remain regarding the long term maintenance of improvements, especially after rapid weight loss with a very low energy diet.12 We evaluated the extent to which initial improvements in the apnoea-hypopnoea index (measured as events/hour) with very low energy diet were maintained after one year in obese patients with moderate to severe obstructive sleep apnoea.
Methods
The study was conducted at the Obesity Unit at Karolinska University Hospital, Karolinska Institute, Stockholm, Sweden, between February 2009 and April 2010.
Participants
The current study is a one year pooled observational follow-up of a previously published randomised trial.10 We included men with moderate to severe obstructive sleep apnoea (apnoea-hypopnoea index ≥15 events/hour) treated with continuous positive airway pressure for a minimum of six months, aged 30-65, and with a body mass index (BMI) of 30-40.2 10
In the controlled trial, 63 patients were randomised to intervention with very low energy diet or a control group for nine weeks. After the randomised phase, the control group crossed over to the same very low energy diet programme as the intervention group. After the very low energy diet period, both groups entered a weight loss maintenance programme. The results for the two groups were pooled for the current analysis (fig 1⇓).
Fig 1 Design and treatment periods in obese men with moderate to severe sleep apnoea
Baseline was defined as the measurement before the start of the very low energy diet (fig 1⇑). Deviations from the inclusion criteria of the randomised controlled trial could therefore occur before crossover in patients randomised to the control group. During the randomised phase the apnoea-hypopnoea index in one patient allocated to control fell to 14 events/hour, and two patients gained weight (to reach a BMI of 40.35 and 40.36). Despite the deviation from the inclusion criteria of the randomised controlled trial, we included these patients in the analysis of the current follow-up study.
Intervention
The full treatment programme of one year consisted of two periods: the very low energy diet period and the weight loss maintenance period.
Very low energy diet period (0-9 weeks)
The very low energy diet weight loss programme consisted of a standard 2.3 MJ/day liquid energy intake protocol (Cambridge Weight Plan, Northants, UK) for seven weeks, followed by two weeks’ gradual introduction of normal food to reach 6.3 MJ/day at week nine. To facilitate compliance with the programme, patients were scheduled for six visits with clinical examinations and group sessions.10
Weight loss maintenance period (9-52 weeks)
The maintenance programme started immediately after the very low energy diet period and was based on standard behaviour modification group therapy with a self help manual.13 14 The programme consisted of three hour group therapy meetings every month. Each group comprised 13-15 patients and was led by a research nurse and a dietitian. In conjunction with the group sessions each patient was seen by a nurse for anthropometry measurements and a dietitian for individual dietary advice.
The behaviour modification focused on nutrition education, eating behaviour, hunger and craving, relapse situations, and increased physical activity. Other important aspects were evaluation of progress and identification of personal and environmental influences affecting eating and physical activity.
To prevent weight regain the protocol specified use of partial meal replacement as a first option (exchanging one or two daily meals with a 0.58 MJ meal replacement, Cambridge Weight Plan, Northants, UK) if the patient’s weight had increased by more than 2 kg since the last visit (86% reported using partial meal replacement at least once). As a secondary option sibutramine or orlistat was prescribed (orlistat was prescribed to only one patient).
Primary outcome
The primary outcome, the apnoea-hypopnoea index, was measured at baseline and nine and 52 weeks. The index is the total number of complete cessations of breathing (apnoea) and partial obstructions (hypopnoea) for at least 10 seconds during sleep.15 The severity was defined according to the recommendations of the American Academy of Sleep, who classify a frequency of 5-14.9 events/hour as mild, 15-30 as moderate, >30 as severe, and <5 as absent.15 In addition to the apnoea-hypopnoea index, we recorded episodes of oxygen desaturation ≥4%/hour of sleep, the nadir of arterial oxygen saturation, and percentage of supine time. Because of night to night variability of sleep studies,16 we derived sleep measurements from two consecutive nocturnal sleep studies in the home, using a six channel ambulatory polygraph (Watch PAT100, Itamar Medical, Caesarea, Israel). We used the Epworth sleepiness scale,17 administered at baseline and nine and 52 weeks, to measure daytime sleepiness. Further details of the sleep assessments can be found elsewhere.10
At one year, the need for continuous positive airway pressure was evaluated at the sleep clinic. Continuous positive airway pressure treatment was terminated if subjective sleep quality and excessive daytime sleepiness were normalised and the apnoea-hypopnoea index was under 10.
Secondary outcomes
Secondary outcomes were body weight, waist and neck circumference, percentage body fat, health related quality of life, metabolic syndrome, hypertension, dyslipidaemia, and insulin resistance (the box shows the definitions used). Metabolic variables were measured at baseline and at nine and 52 weeks according to standard laboratory procedures. Health related quality of life was measured with the SF-12 health survey (to allow quantification of a physical and a mental component score18) and compared with quality of life data for the male Swedish population.19
Definitions used in study
Metabolic syndrome was assessed in accordance with the unified definition of the International Diabetes Federation (IDF) and the American Heart Association/National Heart, Lung, and Blood Institute (AHA/NHLBI), joined by the World Heart Federation, International Atherosclerosis Society, and International Association for the Study of Obesity20
Dyslipidaemia was defined as total cholesterol >5.0 mmol/L, low density lipoprotein >3.0 mmol/L, high density lipoprotein <1.0 mmol/L, and triglycerides >1.7 mmol/L in patients without cardiovascular disease and as total cholesterol >4.5 mmol/L and low density lipoprotein >2.5 mmol/L in patients with cardiovascular disease or receiving drug treatment, or both
Hypertension was defined as systolic/diastolic blood pressure of ≥140/90 mm Hg or greater or receipt of antihypertensive drug treatment, or both
Insulin resistance was calculated with the homoeostasis model assessment (HOMA-IR) (fasting plasma insulin (mU/L)×fasting plasma glucose (mmol/L)/22.5).21 Insulin resistance was defined as HOMA-IR ≥3.9922
Anthropometry and body composition were measured at baseline, after nine weeks, and monthly during the weight maintenance period. Percentage body fat and body weight were measured with a Tanita BC-418MA body fat analyser (Tanita Corporation of America, Arlington Heights, IL).23 Height was measured to the nearest cm with a wall mounted stadiometer. BMI was calculated as weight (kg)/height (m)2. Waist circumference was measured in duplicate halfway between the iliac crest and the lower rib cage with patients standing in underwear. Neck circumference was measured in duplicate at the level of the superior border of the cricothyroid membrane.
Safety
At each visit the study nurse noted adverse events from the very low energy diet, and subsequently the study physician classified these events for potential causality (unlikely/possibly/likely). Previously reported adverse events include dry mouth, diarrhoea, constipation, headache, nausea/vomiting, dizziness, cramps, fatigue, hunger, visual disturbance, feeling cold, hair loss, gallstones, and gout, all of which are considered transient.24
Statistical analysis
Data were analysed with Stata version 10 (College Station, TX) and SPSS (version 17.0, Chicago, IL). Mean values and standard deviations were used unless otherwise stated. Medians and interquartile ranges were used for variables with a skewed distribution.
We used paired samples t test to analyse changes during the very low energy diet period (0-9 weeks), the weight maintenance period (9-52 weeks), and the full treatment programme (0-52 weeks), and independent samples t test to analyse changes in apnoea-hypopnoea index between obstructive sleep apnoea categories at baseline. Correlation and linear regression analysis were used to evaluate predictors of improvement in the apnoea-hypopnoea index and to adjust for potential confounders.
Analyses included all randomised patients. We carried forward baseline observations for missing data to avoid overestimation associated with last observation carried forward because of the rebound in weight that usually occurs after weight loss. Sensitivity analyses for alternative imputation techniques were also performed with the last observation carried forward, multiple imputation, and completers only analysis. For multiple imputation, we used age and baseline value of the variable of interest as explanatory variables. Analysis with multiple imputation and completers should provide similar estimates to the largest effect size, followed by last observation carried forward, and ending with baseline carried forward as the most conservative imputation approach.
To reduce the amount of missing data, we invited the 14 patients who discontinued treatment during weight maintenance to attend the one year follow-up (see appendix 1 on bmj.com). Ten of the 14 (71%) who dropped out completed sleep measurements and six (43%) completed adiposity measurements. Imputation was performed for the five patients who did not start the maintenance programme and the patients who did not attend the one year follow-up. We imputed sleep values in nine and adiposity values in 13.
Results
After crossing over to a very low energy diet, the patients who had served as controls in the randomised phase10 achieved the same weight loss (−20 v −19 kg; mean difference −1 kg, 95% confidence interval −3 to 1; P=0.34) and improvement in the apnoea-hypopnoea index (−21 v −25 events/hour; mean difference −4, −12 to 5; P=0.38) as the first intervention group. The finding of greater improvements in patients with severe versus moderate disease10 was also replicated after crossover of the controls (−26 v −13 events/hour, P=0.02).
By pooling the two intervention groups, 58 of 63 patients completed the very low energy diet period (two patients never started and three dropped out). At one year 49 patients had completed sleep and adiposity follow-up measures, and 44 had completed the one year treatment (see appendix 1 on bmj.com).
At baseline the majority of patients had severe obstructive sleep apnoea, metabolic syndrome, hypertension, and dyslipidaemia (table 1⇓, and 56%, 41%, and 3% had a BMI of 30-34.9, 35-39.9, and ≥40, respectively. Those with moderate and severe obstructive sleep apnoea had a mean apnoea-hypopnoea index of 23 and 46 events/hour, respectively. The physical quality of life component (as measured with SF-12) was lower than in the general male Swedish population19 (47 v 51 units, P<0.001), while the mental component was similar (53 v 54 units, P=0.57).
Baseline characteristics of obese men with moderate to severe obstructive sleep apnoea
Changes in adiposity
During the very low energy diet and full treatment programme, weight, BMI, waist circumference, neck circumference, and percentage body fat all decreased significantly, whereas all adiposity variables increased significantly during the weight maintenance period (table 2⇓ and fig 2⇓).
Fig 2 Mean change from baseline in weight, waist circumference, neck circumference, and percentage body fat during and after treatment with very low energy diet for patients completing weight loss maintenance programme (n=44) and sensitivity analysis for missing data with multiple imputation (n=63), last observation carried forward (LOCF; n=63), or baseline observation carried forward (BOCF; n=63). Attendance was low at 15 and 23 weeks because of summer holidays. Error bars indicate 95% confidence intervals
Changes in anthropometry (adiposity) after low energy diet and maintenance programme in 63 obese men with moderate to severe sleep apnoea (baseline observation carried forward for missing data), with P values from paired sample t test
Of the 63 participants analysed at the one year follow-up, one man (2%, 95% confidence interval −1% to 5%) was normal weight (BMI <25), 27 (43%, 31% to 55%) were overweight (BMI 25.0-29.9), and 35 (56%, 43% to 68%) remained obese (BMI ≥30).
Changes in obstructive sleep apnoea
All sleep variables, including the apnoea-hypopnoea index, improved significantly after the very low energy diet period and full treatment programme (table 3,⇓ fig 3,⇓ and appendix 4 on bmj.com). During the weight maintenance period the apnoea-hypopnoea index and oxygen desaturation index increased significantly (table 3; see appendix 2 on bmj.com for sensitivity analyses with different imputation methods). At the one year follow-up, six (10%, 95% confidence interval, 2% to 17%) had total remission of obstructive sleep apnoea (apnoea-hypopnoea index <5), and 30 (48%, 35% to 60%) patients no longer required continuous positive airway pressure, of whom 23 did not need any further treatment with seven shifted to treatment with an oral appliance (an orthodontic appliance worn in the mouth during sleep that keeps airways free from obstruction).
Fig 3 Proportions with obstructive sleep apnoea at baseline and after treatment with very low energy diet and after one year in 63 participants (baseline observation carried forward for missing data). Apnoea-hypopnoea index: remission <5, mild=5-14.9, moderate=15-30, severe obstructive sleep apnoea >30 events/hour
Changes in sleep variables after low energy diet and maintenance programme in 63 obese men with moderate to severe sleep apnoea (baseline observation carried forward for missing data), with P values from paired sample t test
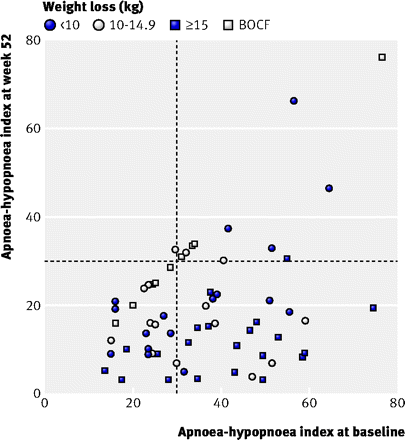
Improvements in the apnoea-hypopnoea index were larger in men with severe obstructive sleep apnoea at baseline than in those with moderate disease (−25 v −7 events/hour, P<0.001). Indeed, at one year there was no difference in the apnoea-hypopnoea index between patients who started with severe compared with moderate disease (22 v 16 events/hour, P=0.09; fig 4⇓).
Fig 4 Association between apnoea-hypopnoea index at baseline and follow-up for intention to treat population (BOCF=baseline observation carried forward) and weight loss categories in 63 participants. Dashed line represents cut off for severe sleep apnoea (index >30 events/hour)
Dose-response of weight loss and change in obstructive sleep apnoea
A dose-response relation existed between changes in adiposity and apnoea-hypopnoea index: body weight (r=0.41, P=0.004), waist circumference (r=0.40, P=0.005), neck circumference (r=0.33, P=0.02), and percentage body fat (r=0.40, P=0.004), fig 5⇓.
Fig 5 Association between apnoea-hypopnoea index and weight loss (kg). Top: relation between apnoea-hypopnoea index and weight change in all 63 participants. Correlation coefficient and regression line based only on those with measured weight and index at baseline and follow-up (n=49). Bottom: relation between apnoea-hypopnoea index and weight for all participants with measured weight and index at baseline and follow-up (n=49). Lines indicate individuals. No participant gained weight, meaning that right-most observation represents baseline value
Patients who lost ≥15 kg had larger improvements in the apnoea-hypopnoea index at one year than patients who lost 10-14.9 kg (−30 v −15, P=0.004) or those who lost <10 kg (−30 v −15, P=0.008).
After adjustment for baseline age, weight, apnoea-hypopnoea index, and allocation (first or second very low energy diet group), weight loss was significantly associated with apnoea-hypopnoea index at follow-up (β=0.50 events/kg, 0.11 to 0.88; P=0.013)—that is, a 10 kg weight loss was associated with an average decrease of 5 events/hour in the apnoea-hypopneoa index at follow-up (see appendix 3 on bmj.com). Patients who regained weight during the weight maintenance period had a significantly higher mean apnoea-hypopnoea index at follow-up (β=0.48 events/kg, 0.08 to 0.87; P=0.02) after adjustment.
Quality of life
During the very low energy diet period, the physical quality of life component increased by 5 units (3 to 7; P<0.001), while the mental component was unchanged (2, −0.4 to 4; P=0.11) and remained similar to the level in the general population.19 Between baseline and one year follow-up, the physical component score had increased by 4 units (2 to 6; P<0.001) and was similar to that in the general population (50 v 51; P=0.73).
Metabolic risk
Between baseline and one year follow-up all metabolic variables in table 1 improved significantly. Among patients with metabolic disease at baseline, dyslipidaemia disappeared in 11/59 (19%, 9% to 29%), insulin resistance in 10/20 (50%, 28% to 72%), and metabolic syndrome in 23/44 (52%, 38% to 67%).
There were no differences in either systolic (−2 mm Hg, −6 mm Hg to 2 mm Hg; P=0.36) or diastolic blood pressure (−1 mm Hg,−1 mm Hg to 3 mm Hg; P=0.48) during the full treatment programme. Of those with hypertension at baseline, however, it had disappeared in 22% (8/36).
Adverse events
During the very low energy diet period 13/63 patients (21%, 11% to 31%) had an adverse event classified as probably causally linked with the very low energy diet. These adverse events were constipation (n=3), increased alanine aminotransferase activity (n=6), dizziness (n=1), gout (n=2), and dry lips (n=1). All adverse events had disappeared by the visit two weeks after the very low energy diet period.
During weight loss maintenance there were five additional adverse events (5/58, 9%, 1% to 16%) probably causally linked to treatment with very low energy diet (gallstones (n=3), gout (n=1), and kidney stones (n=1)). No patient discontinued treatment because of adverse events.
Discussion
In this prospective observational study in obese men with obstructive sleep apnoea, changes in the apnoea-hypopnoea index after nine weeks of a very low energy diet (−58%) were largely maintained at one year (−47%). Patients with severe obstructive sleep apnoea at baseline had larger improvements in the apnoea-hypopnoea index than patients with moderate disease (−25 v −7 events/hour). Moreover, we found a dose-response association between weight loss and improvement in the apnoea-hypopnoea index, with patients who lost the most improving the most. At follow-up, 30 (48%) men no longer required continuous positive airway pressure, and six (10%) had total remission of obstructive sleep apnoea. There were also marked improvements in metabolic risk and the physical dimension of quality of life, which no longer differed from the level in the general population.
Previous research
Randomised controlled trials have previously shown a positive effect of weight loss on obstructive sleep apnoea. Tuomilehto et al found a 47% reduction in the apnoea-hypopnoea index in patients with mild obstructive sleep apnoea after a very low energy diet (at 12 weeks), a 40% reduction with 11 kg weight loss after one year, and a 46% reduction with a 7 kg weight loss after two years.8 25 Foster et al found a 24% reduction in the apnoea-hypopnoea index after one year with 11 kg weight loss after an intensive lifestyle intervention in older (mean age 61) patients with type 2 diabetes, while the apnoea-hypopnoea index increased in control patients, resulting in a between group difference of 42%.9 Finally, the randomised part of our study documented a 67% reduction in the apnoea-hypopnoea index after nine weeks after a mean weight loss of 19 kg in middle aged men with moderate to severe disease.10 The dose-response association we found between weight loss and the apnoea-hypopnoea index was also found by Foster et al9 and Tuomilehto et al8 25 and in our previous study.10 Foster et al also found significantly greater improvements in patients with severe disease at baseline,9 as in the current and our previous study.10
In addition to randomised controlled trials, two uncontrolled studies,26 27 with smaller sample sizes (n=24-33) and complete case analysis, have investigated maintenance of initial improvements in obstructive sleep apnoea after a very low energy diet. Both Lojander et al26 and Kajaste et al27 found that obstructive sleep apnoea was improved or largely maintained after one year in patients completing the study.
Our study extends previous findings by highlighting the long term benefits of weight loss in people with moderate and especially severe obstructive sleep apnoea, which have both been associated with increased mortality,3 4 11 in contrast with mild sleep apnoea investigated previously by Tuomilehto et al.8 25
In addition to dietary interventions, weight loss surgery has also been shown to result in long term improvements in obstructive sleep apnoea. In a large study (n=3477) including matched controls, Grunstein et al found that 30% of the surgery group reported persistent sleep apnoea at the two year follow-up compared with 70% of the control group.28 Small uncontrolled studies (n<20) have also found long term improvements in obstructive sleep apnoea after weight loss surgery.29 30 31 32
Clinical implications
According to previous studies, weight loss programmes should ideally achieve a weight loss of at least 10-15 kg.8 9 25 Very low energy diet treatment followed by a weight loss maintenance programme is one option for achieving long term weight loss of such magnitude.33 Three other studies have found that improvements in obstructive sleep apnoea in other patient groups were maintained one8 9 and two years after very low energy diet.25
A very low energy diet is associated with some adverse events, notably gallstones. There is no reason to believe that these well known side effects occur differently in patients with sleep apnoea than in obese patients in general. The risk of adverse events, however, needs to be considered by clinicians and patients before treatment is started.
Weight loss trials tend to be afflicted by high levels of attrition with reported dropout levels of 30-39%.34 In the current study 30% (19/63) of participants did not complete the full treatment programme, while 14% (9/63) were unavailable for the sleep study at follow-up. The current study therefore has a similar proportion of patients not assessed at end of the study as in the studies by Tuomilehto et al (11%)8 and Foster at al (17%).9
Strengths and weaknesses
The strengths of the current study include the duplicated sleep studies, the focus on patients with increased risk of mortality, and a design that allowed analysis of whether initial improvements in obstructive sleep apnoea can be maintained after rapid weight loss induced by a very low energy diet.
The main limitation is the observational study design. The reason for not using a randomised design with a one year follow-up was that our primary aim was to show, in the short term, a treatment effect from weight loss with a very low energy diet in moderate to severe obstructive sleep apnoea. Our secondary aim was to see whether any improvements could be maintained in the long term. We therefore wanted to minimise non-compliance in the control group in the nine week randomised phase by providing a strong incentive for controls to remain in the study by allowing patients to start the very low energy diet programme immediately after serving as controls.
The observational design means that our analysis is limited by the lack of comparison with natural progression in the apnoea-hypopnoea index and potential regression to the mean. Without a control group, we might have underestimated the one year treatment effect, as Foster et al showed an increase in apnoea-hypopnoea index in their control group over one year.9 The lack of control group also makes it difficult to investigate if the larger reduction in the apnoea-hypopnoea index in patients with severe rather than moderate disease at baseline is a result of regression to the mean rather than a true treatment effect. The effect modification by baseline apnoea-hypopnoea index, however, was seen both in the randomised part of this study with the same population10 and in the study by Foster et al,9 suggesting it is not purely caused by regression to the mean.
Another limitation was the use of a portable WatchPAT device instead of a polysomnograph, which is considered the ideal for diagnosing obstructive sleep apnoea. The portable WatchPat does not technically measure airflow but quantifies autonomic consequences of disordered breathing events during sleep and is therefore considered a close surrogate measure of the apnoea-hypopnoea index as defined by the American Academy of Sleep.15 Validation studies show that the WatchPAT device has high sensitivity and specificity for pathological respiratory disturbances, estimating sleep time and apnoea-hypopnoea index accurately compared with polysomnography.35 36
Finally, our results might not be generalisable to women, younger (<30) or older (>65) patients, overweight (BMI 25-29.9) or extremely obese patients (BMI ≥40), or patients with mild sleep apnoea.
Conclusions
Improvements in apnoea-hypopnoea index after weight loss induced by a very low energy diet were largely maintained after one year, with patients with severe obstructive sleep apnoea experiencing larger reductions in the apnoea-hypopnoea index than those with moderate disease at baseline. There was also a dose-response association between weight loss and improvement in the apnoea-hypopnoea index, with patients who lost the most improving the most.
What is already known on this topic
Obstructive sleep apnoea is associated with several adverse outcomes, including impaired cognitive function, vehicle crashes and occupational injuries, and death
Randomised controlled trials have recently shown that weight loss improves obstructive sleep apnoea in overweight and obese patients
The long term effect of weight loss has been studied only in people with mild obstructive sleep apnoea and in older patients with type 2 diabetes
What this study adds
The initial improvements in apnoea-hypopnoea index after nine weeks of a very low energy diet (−58%) were largely maintained at the one year follow-up (−47%)
At one year, 48% (30/63) no longer required continuous positive airway pressure and 10% (six/63) had total remission of obstructive sleep apnoea
Patients who lost the most in weight or had severe sleep apnoea at baseline benefited most
Notes
Cite this as: BMJ 2011;342:d3017
Footnotes
We thank the study nurse Lena Mannström and the dietitians Jenny Dygve and Mary Hyll for treating all patients, and the nurses Anna Laumann, Viveca Petré, and Sara Yllö for assistance during the study.
Contributors: KJ, MN, SR, and EH conceived the study hypothesis. SR and YTL met all the patients, RH conducted the sleep studies, and KJ and MN conducted the statistical analyses. KJ wrote the first draft of the manuscript. MN, EH, SR, YTL, RH, and FG critically reviewed and contributed to the final draft. All authors are guarantors.
Funding: This study was partly supported by research grants from Cambridge Weight Plan, Northants, UK, and Novo Nordisk AS, Bagsværd, Denmark. No one representing the funding sources read or commented on any version of the manuscript.
Competing interests: All authors have completed the Unified Competing Interest form at www.icmje.org/coi_disclosure.pdf (available on request from the corresponding author) and declare: KJ, SR, and EH have received travel grants from Cambridge Weight Plan to attend a scientific meeting; no other relationships or activities that could appear to have influenced the submitted work.
Ethical approval: This study was approved by the regional ethics committee in Stockholm, Sweden. Written informed consent was obtained from all patients.
Data sharing: No additional data available.
This is an open-access article distributed under the terms of the Creative Commons Attribution Non-commercial License, which permits use, distribution, and reproduction in any medium, provided the original work is properly cited, the use is non commercial and is otherwise in compliance with the license. See: http://creativecommons.org/licenses/by-nc/2.0/ and http://creativecommons.org/licenses/by-nc/2.0/legalcode.