Cost effectiveness of an intensive blood glucose control policy in patients with type 2 diabetes: economic analysis alongside randomised controlled trial (UKPDS 41)
BMJ 2000; 320 doi: https://doi.org/10.1136/bmj.320.7246.1373 (Published 20 May 2000) Cite this as: BMJ 2000;320:1373- Alastair Gray, director (alastair.gray{at}ihs.ox.ac.uk)a,
- Maria Raikou, research training fellowb,
- Alistair McGuire, economistb,
- Paul Fenn, economistc,
- Richard Stevens, statistical modellerd,
- Carole Cull, senior statisticiand,
- Irene Stratton, senior statisticiand,
- Amanda Adler, epidemiologistd,
- Rury Holman, directore,
- Robert Turner, directord the United Kingdom Prospective Diabetes Study Group
- a Health Economics Research Centre, Department of Public Health, University of Oxford, Institute of Health Sciences, Headington OX3 7LF
- b Department of Economics, City University, London EC1V 0HB
- c Business School, University of Nottingham, Nottingham NG7 2RD
- d Diabetes Research Laboratories, Nuffield Department of Clinical Medicine, University of Oxford, Oxford OX2 6HE
- e Diabetes Trials Unit, Nuffield Department of Clinical Medicine
- Correspondence to: A Gray
- Accepted 7 February 2000
Abstract
Objective: To estimate the cost effectiveness of conventional versus intensive blood glucose control in patients with type 2 diabetes.
Design: Incremental cost effectiveness analysis alongside randomised controlled trial.
Setting: 23 UK hospital clinic based study centres.
Participants: 3867 patients with newly diagnosed type 2 diabetes (mean age 53 years).
Interventions: Conventional (primarily diet) glucose control policy versus intensive control policy with a sulphonylurea or insulin.
Main outcome measures: Incremental cost per event-free year gained within the trial period.
Results: Intensive glucose control increased trial treatment costs by £695 (95% confidence interval £555 to £836) per patient but reduced the cost of complications by £957 (£233 to £1681) compared with conventional management. If standard practice visit patterns were assumed rather than trial conditions, the incremental cost of intensive management was £478 (-£275 to £1232) per patient. The within trial event-free time gained in the intensive group was 0.60 (0.12 to 1.10) years and the lifetime gain 1.14 (0.69 to 1.61) years. The incremental cost per event-free year gained was £1166 (costs and effects discounted at 6% a year) and £563 (costs discounted at 6% a year and effects not discounted).
Conclusions: Intensive blood glucose control in patients with type 2 diabetes significantly increased treatment costs but substantially reduced the cost of complications and increased the time free of complications.
Introduction
Improved blood glucose control is known to decrease progression of microvascular disease in patients with type 1 diabetes, and the cost effectiveness of this policy has been reported using data from the diabetes control and complications trial.1 Without information on clinical treatments and their long term impact on disease progression it has not been possible to assess the cost effectiveness of similar strategies in patients with type 2 diabetes. Previous economic evaluations have used existing knowledge of the disease epidemiology to consider specific aspects of disease progression such as retinopathy.2 3 Model-based evaluations have also been reported, the most inclusive of which predicted rates of microvascular complications, cardiovascular disease, and mortality.4 5 The United Kingdom prospective diabetes study provides, for the first time, the necessary clinical information on both microvascular and macrovascular complications to allow the cost effectiveness of an improved glucose control policy in people with type 2 diabetes to be analysed. The median 10 year follow up in the study makes it possible to estimate long term resource implications of type 2 diabetes and its complications directly from trial data.6
Methods
Participants and comparisons
A total of 5102 newly diagnosed patients with type 2 diabetes, defined as fasting plasma glucose above 6 mmol/l on two occasions, aged 25–65 years (mean age 53) were recruited in 23 centres. After initial dietary treatment, 4209 patients had fasting plasma glucose concentrations of 6.1-15 mmol/l without symptoms of hyperglycaemia. Of these, 342 overweight patients were randomised to metformin, leaving 3867 patients who entered the main randomisation and were allocated either to conventional management (mainly through diet, 1138 patients), or to intensive management with insulin (1156) or sulphonylureas (1573). The aim of the conventional policy was to maintain patients free of diabetic symptoms and with a fasting plasma glucose concentration below 15 mmol/l, whereas the intensive policy was aimed at a fasting plasma glucose concentration below 6 mmol/l. All patients in the main randomisation were included in this economic evaluation. The median follow up period to death, the last known date at which survival was known, or to the end of the trial was 10 years. The main clinical end points analysed were death or the development of diabetic complications, including coronary heart disease, cerebrovascular disease, amputation, laser treatment for retinopathy, cataract extraction, and renal failure. All analyses and comparisons were performed on an intention to treat basis.
Type of evaluation and perspective
We performed an incremental cost effectiveness analysis in which the net costs and net effectiveness of intensive compared with conventional management were calculated and expressed as a ratio. The main perspective of the economic evaluation was that of healthcare purchasers. Only direct health service costs were analysed. These costs covered conventional and intensive treatments, visits to diabetic clinics and tests, and treatment of diabetic complications, including inpatient stays and outpatient health care. We also compared the costs of conventional policy with the insulin and sulphonylurea intensive policies separately.
Resource data
For each patient, data were collected at three monthly clinic visits on the doses of all drugs used for treating diabetes (insulin, sulphonylureas, metformin); the number of home blood glucose tests; the dose of the three main drugs for hypertension (captopril, atenolol, nifedipine); whether the patient was taking diuretics, methyldopa, calcium channel blockers, vasodilators, or other antihypertensive drugs; and whether the patient was taking aspirin, antidepressant drugs, hormone replacement therapy, anxiolytics, or any other drugs. When treatment doses were not recorded, missing values were replaced by extrapolation from adjacent values for that patient. Last observation carried forward was used to impute missing data when necessary.
Data on the date and duration of each hospital admission were collected at every clinic visit. These were coded by using ICD-9 and ICD-10 classifications for prime cause of admission and Office of Population Censuses and Surveys (OPCS-4) codes for all procedures undertaken. In addition, a separate record was maintained of all angiograms, angioplasties, and bypass grafts for coronary or peripheral vascular disease. All hospital admissions were also allocated by two clinicians to one of 40 national standard specialty codes. Missing values for hospital lengths of stay were replaced with the mean value for all patients in that specialty.
Data on non-hospital and outpatient resource use were collected from all patients in the trial through a questionnaire distributed at routine clinic visits between January 1996 and September 1997 and by post to those who did not attend a clinic during this period. This questionnaire collected information on all home, clinic, and telephone contacts with general practitioners, nurses, chiropodists, opticians, dieticians, and eye and other specialists over the previous four months. Retrospective data capture from patients may underestimate resource use, but this is unlikely to introduce systematic bias when analysed by allocation. These cross sectional data were analysed by multiple regression to estimate for each patient the annual non-hospital resource use adjusted for significant variables including age, sex, body mass index, duration of diabetes, and time from a non-fatal diabetes related end point.
Costs
Unit costs for all resources used by trial patients were obtained from national statistics and from centres participating in the trial (table 1). These unit costs were combined with the resource volumes to obtain a net cost per patient over their time in the trial. Mean net costs and associated 95% confidence intervals were calculated for each arm of the study. Costs are reported both undiscounted and in net present values using the UK Treasury approved 6% annual discount rate.8 All costs are reported in 1997 values (£s).
Main unit costs and sources of conventional and intensive management of type 2 diabetes
All participants in the study attended clinics every three months; the interval was increased to four months in the later years of the study. However, outside a trial it is likely that the frequency and type of visits would be different, particularly for conventional policy patients. To illustrate this, we conducted a complementary analysis in which visits for conventional or intensive treatment are costed to reflect likely standard clinical practice rather than that dictated by the trial protocol. This assumes that the observed differences in complications between trial arms would be maintained with the different pattern of visits. table 2 outlines the likely pattern of standard practice for conventional and intensive management based on the opinion of primary care and specialist clinic staff in the participating centres. Each patient's actual annual trial visit costs were replaced by the estimated standard practice annual visit cost depending on allocation and the associated probability of the patient receiving insulin or diet and tablet treatment. The costs of other patterns are also considered in sensitivity analyses.
Assumed annual real life visit and blood glucose test schedules for conventional and intensive management of type 2 diabetes
Outcomes
Diabetes related end points were defined as in the clinical trial.6 The trial showed that intensive blood glucose control significantly reduced (P=0.029) the risk of any diabetes related end point by 12% but did not significantly reduce diabetes related deaths or all cause mortality. Consequently, the current analysis measures outcomes in terms of time to first event (myocardial infarction, congestive heart failure, stroke, renal replacement therapy, amputation, cataract extraction, vitreous haemorrhage, or death from any cause).
We used a conservative estimate of time to first event—that is, we assumed that no treatment effects continue beyond the end of the trial. For patients with no event observed during the trial, we used simulation from a parametric model to estimate the time from study closure (or withdrawal from study) to first event. Basic bootstrap confidence intervals were calculated for all simulation results based on 5000 non-parametric bootstrap iterations, each iteration being averaged over 20 runs of the simulation for stability.
For comparison we also estimated time to first event assuming that treatment effects continue beyond the end of the trial. We refer to this as an unbiased estimate, because it attempts to follow the size of the treatment effects that were observed in the trial, whereas the conservative estimate described above forces all treatment effects to zero at study closure. All cost effectiveness results are based on the conservative estimate of effectiveness. Since cost data are available for the duration of the trial only, the appropriate effectiveness measure is the one that allows treatment effects during the trial only.
Models
The model used in the simulation described above was a twofold competing risk model. In the first component, risk of a diabetes related event increases with age at diagnosis of diabetes and with duration of diabetes. In the second component, risk of other death (any death except myocardial infarction, sudden cardiac death, stroke) increases with the age of the patient. For simplicity, the last category includes several causes of death that may be considered diabetes related (hyperglycaemic or hypoglycaemic episodes, renal death, and death from peripheral vascular disease) but occurred too infrequently to be modelled individually (total 26 deaths). The methods used to test the model's validity are given on the BMJ's website.
Analysis
All comparisons were carried out on an intention to treat basis. All results are reported as mean values with standard deviations; mean differences are reported with 95% confidence intervals. When descriptive statistics suggested the possible presence of skewness, 1000 bootstrap replications of the original data were performed and the resulting means, mean differences, and intervals were compared. For all reported costs, parametric confidence intervals for the cost differences were compared with the bootstrap confidence intervals and were found to be robust; parametric confidence intervals are therefore reported. Confidence intervals for the mean cost effectiveness ratios were calculated by Fieller's method.9 10 The effect of assumptions on our main results was examined by sensitivity analyses. All data were analysed with SPSS 8.0 and Microsoft Excel 97; the modelling work was carried out in C language.
Results
Table 3 shows the associated mean cost per patient over the duration of the study by category of cost and allocation. The intensive glucose control policy increased the costs of antidiabetic treatment for each patient by an average of £659 (95% confidence interval £580 to £739) compared with conventional glucose control. There were no significant differences between patients in the conventional and intensive glucose control policy groups in the costs of antihypertensive drugs, other drugs, or trial clinic visits. Total routine treatment costs were £3655 per patient in the conventional group and £4350 in the intensive group (mean difference £695, 95% confidence interval £555 to £836). When trial visit and test costs were replaced by the estimates of standard clinical practice visit and test patterns, as shown in table 2, total treatment costs were £1658 in the conventional group and £3091 in the intensive group (mean difference £1435, £1332 to £1539).
Mean costs and effects for intensive and conventional blood glucose control policies
Complication costs
Hospital admissions formed the largest element of complication costs (table 3). The mean cost of all hospital admissions was £4266 in the conventional group and £3494 in the intensive group (mean difference £772, £159 to £1385). The 18% reduction in cost with intensive management is primarily the result of differences in the length of stay (9.7 days in conventional group v 8.4 in intensive group; mean difference 1.3, 0.2 to 2.3) combined with small differences in the number of admissions (mean 1.5 episodes in conventional group v 1.4 in intensive group; mean difference 0.1 (−0.1 to 0.2, not significant)).
Cross sectional analysis of responses to the questionnaire on non-hospital and outpatient healthcare use indicated that a recent end point event had a significant effect on costs, raising them on average by £241 in the first year, £106 in the second year, and £80 in the third year after the event. Thus a lower event rate in the intensive group should be associated with lower non-hospital costs. Over the trial period, costs were slightly lower in the intensive group but the difference was not significant. Similarly, the costs associated with treatment of eye and renal disease (primarily renal dialysis and retinal photocoagulation) were slightly but not significantly lower in the intensive group. In total, therefore, intensive management was associated with a reduction of £957 (£233 to £1681) per patient in the cost of complications compared with conventional policy.
Total costs
The increased costs of antidiabetic treatment among the intensive group were counterbalanced by reduced costs of complications so that the net trial costs per patient did not differ between the two groups (£9869 in the conventional group and £9608 in the intensive group). Discounted at 6% a year to present values these costs become £7170 in the conventional group and £6958 in the intensive group.
However, when the trial visits and tests were replaced by those likely in clinical practice (table 2), the cost per patient was £7871 for conventional management v £8349 for intensive management (mean difference £478, −£275 to £1232). This difference is not significant. Discounted at 6% a year to present values the costs become £5689 v £6027 (£338, −£207 to £882). Comparisons of costs per patient for conventional treatment with insulin and sulphonylurea treated groups separately showed no signifcant differences (see BMJ's website for details).
Costs over time
The costs reported above are aggregated per patient over the whole trial period. Because of the nature of the disease costs will increase over time. To illustrate this, figure 1 shows the mean undiscounted costs per patient by year from their date of randomisation. There were no significant differences in the mean treatment costs per patient between conventional and intensive patients when considering antihypertensive drugs, other drugs, or trial clinic visits. Although we found some differences in the costs associated with the treatment of eye and kidney disease over time, these differences arose from such a small number of events that, as indicated in table 3, the mean cost difference per patient over the whole trial was not significant. These are therefore not included in figure 1.
Mean cost per patient by year from randomisation and allocation to conventional and intensive glucose control policies based on resource use in trial (undiscounted costs in 1997 £).
Outcomes
The main measure of effectiveness in this analysis is time to first event. The conservative estimate was 14.29 years in the conventional group and 14.89 years in the intensive group, a difference of 0.60 (0.12 to 1.10) years (table 3). Discounted to present values at 6% a year, mean time to event was 8.88 years in the conventional group and 9.17 years in the intensive group (0.29, 0.06 to 0.53 years).
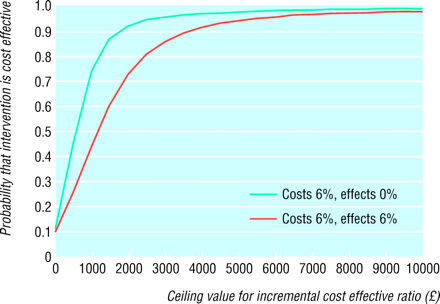
Cost effectiveness acceptability curves: probability that the within trial cost per event-free year gained is cost effective (y axis) as a function of decision maker's ceiling cost effectiveness ratio (x axis)
The unbiased model in which treatment effects are assumed to continue beyond the trial estimates a mean gain of 1.14 (0.69 to 1.61) years for intensive management compared with conventional management. The conservative estimate is used in the analysis below.
Cost effectiveness
The primary measure of cost effectiveness is the incremental cost per event-free year gained. Discounting both costs and effects to present values at 6% a year, the intervention is more effective and cost saving based on resource use according to the trial protocol. If the standard practice volumes are used rather than the protocol driven volumes the cost per event-free year gained, with both costs and effects discounted at 6%, is £1166 (−£692 to £8819). With the costs discounted at 6% a year and the effects undiscounted, the cost per event-free year gained is £563 (−£344 to £5632).
Figure 2 shows these results in the form of a cost effectiveness acceptability curve.11 With costs and effects discounted at 6% a year, there is a 10% probability that intensive blood glucose control policy would prove to be cost saving compared with a conventional policy, a 50% probability that the cost per event-free year lies above (or below)the point estimate of £1166, and an 80% probability that the ratio is less than £2500.
Sensitivity analysis
Sensitivity analysis was performed on the cost effectiveness ratio resulting from the main analysis, in which both the incremental net costs and incremental effect were discounted at 6% a year and the study visits reflected standard practice. The analysis focused on variation in the likely pattern and cost of visits and blood glucose test schedules in standard practice from the baseline values in table 2. If the frequency of the visits to a doctor at a diabetes hospital clinic increased from once to twice a year for the intensive policy group treated with insulin and from 0.5 to once a year for the intensive policy group treated with oral drugs, the difference in cost between the intensive and conventional policies would become £625 (£81 to £1168), and the incremental cost per event-free year would increase from £1166 to £2155. If the frequency of the visits to a specialist nurse increased from once to twice a year for the conventional policy group receiving insulin and from none to once a year for the conventional policy group receiving oral drugs, the difference in the cost between the policies would be £165 (−£379 to £710). The resulting incremental cost per event-free year would decrease to £572.
Discussion
Our economic analysis of treatment options in blood glucose control for people with type 2 diabetes is based directly on clinical trial information. The data are therefore less prone to the sources of bias, confounding, and uncertainty that are likely to affect non-randomised study designs. Secondly, because of the long follow up in the UK prospective diabetes study, the full range of costs arising from diabetic complications under conventional and intensive management could be assessed empirically. The relation between glycaemia and outcome is complex, but the UK prospective diabetes study has shown that improved glucose control reduces the risk of the diabetic complications that cause morbidity and suggested the mechanisms by which this might occur.6
Our economic analysis shows that the additional costs of intensive management are largely offset by significant reductions in the costs of treating complications of diabetes. If the prevalence of type 2 diabetes is 1.0-1.9% in the general population, a practice with a list of 10 000 patients will typically have 100–190 patients with type 2 diabetes at any time. Under our assumed clinical conditions an intensive policy costs an additional £1435 per patient (about £140 a year), which would be £14 000-£27 000 for a practice. These costs would be offset by £10 000-£18 000 in savings on complications.
Further research
Further evaluations will be needed to examine different ways in which an intensive blood glucose control policy can be translated into standard practice and the role of new drugs. Future studies could also cast further light on the non-hospital costs of diabetic complications, which we assessed using cross sectional data obtained towards the end of the study. We did not include any potential difference between trial groups in productivity losses to individuals and society or costs directly incurred by patients or their families. As intensive blood glucose control was associated with shorter hospital admissions, it may result in fewer such indirect costs.
What is already known on this topic
Intensive blood glucose control in patients with type 2 diabetes significantly reduces the risk of diabetes related complications
Intensive control has been shown cost effective in type 1 diabetes but data are lacking for type 2 diabetes
What this study adds
The increased therapy costs of intensive blood glucose control in type 2 diabetes are largely offset by significantly reduced costs of complications
In a typical general practice, the net cost of intensive blood glucose control for all type 2 diabetic patients is likely to be £4000-£9000 annually
The cost per event-free year of intensive blood glucose control is about £1166
Given the wide range of complications associated with diabetes, it is important to attempt to define a measure that captures all dimensions of health gain. This is particularly true when considering a preventive treatment that delays an individual's progression to illness. Event-free time is one composite measure of health that reflects the complex nature of outcome. However, event-free time does not capture fully all the potential health effects. In particular, it does not incorporate information on quality of life. It might be expected that individuals receiving intensive glucose control who experience a gain in event-free time would consequently see an improvement in their quality of life. The intensive policy itself does not seem to adversely affect quality of life, although more hypoglycaemic events and weight gain may occur.12 Our focus on event-free years allowed us to use the same outcome measures used in the UK prospective diabetes study.6
In principle economists are interested in lifetime costs and effects, which may differ substantially from costs and effects within trials. In a trial such as the United Kingdom prospective diabetes study with long follow up this is less likely to be a serious issue. The results reported in this analysis relate purely to the costs and treatment effects observed within the trial period and do not require extrapolation of effects beyond the trial period. However, modelling indicated that these longer term benefits are likely to double the mean difference in event-free time. Our results suggest that intensive management of patients with type 2 diabetes is a feasible and economically supportable option.
Acknowledgments
We thank the patients and staff at the study centres for their cooperation and Andrew Briggs, Philip Bassett, Valeria Frighi, and Ziyah Mehta for their contributions.
Contributors: AG helped plan and design the health economics study, supervised and participated in the analysis, coordinated the writing of the paper, and is the guarantor. MR compiled, maintained, and analysed the economic data and participated in interpreting the results and writing the paper. AMcG helped plan and design the health economics study and participated in supervising analysis, interpreting results, and writing the paper. PF helped plan and design the health economics study and participated in supervision and interpretation of results and revising the paper. RS developed the simulation model and participated in interpreting the results and writing the paper. CC was responsible for maintenance of the database, prepared trial data for analysis, discussed statistical aspects of the analysis, and participated in revising the paper. IS participated in data preparation, interpretation, and revision of the paper. AA helped to develop the simulation model and participated in interpreting the results and revising the paper. RH was a principal clinical investigator in the UK prospective diabetes study and participated in data preparation, analysis, and interpretation and revising the paper. RT was a principal clinical investigator in the clinical study, helped plan and design the study, and participated in interpreting the results and initial drafting of the paper. He died in August 1999.
Footnotes
-
Funding Glaxo Wellcome, SmithKline Beecham, Pfizer, Zeneca, Pharmacia Upjohn, Novo Nordisk, Bayer, Roche, and UK Department of Health. RS is supported by a Wellcome Trust fellowship (grant No 054470/Z/98/Z/DG/NOS/FH). The main study was supported by grants from the UK Medical Research Council, British Diabetic Association, UK Department of Health, US National Eye Institute and National Institute of Diabetes, Digestive and Kidney Disease in the National Institutes of Health, British Heart Foundation, Novo Nordisk, Bayer, Bristol-Myers Squibb, Hoechst, Lilly, Lipha and Farmitalia Carlo Erba.
-
Competing interests RH has received research for members of staff and fees for consulting and speaking, CC has received support for the cost of attending conferences, and AA has received fees for consulting from many of the companies who supported the study. AG has received support for attending conferences from Lipha.
-
 The centres of the UK Prospective Diabetes Study Group, methods used to test model validity, and results of secondary analysis are given on the BMJ's website
The centres of the UK Prospective Diabetes Study Group, methods used to test model validity, and results of secondary analysis are given on the BMJ's website